ESKD in IgAN: A clinical consequence that cannot be overlooked1,2
IgA Nephropathy (IgAN), a leading cause of end-stage kidney disease (ESKD), requires a treatment that moves beyond symptom control to target the origin of pathophysiology and halt disease progression.1,3
All patients with IgAN are at risk of kidney failure within their lifetime1
IgAN is a chronic, B-cell–mediated, autoimmune kidney disease characterized by a progressive, irreversible decline in kidney function, high rates of kidney failure, and an increased risk of early death.2,3
IgAN can affect patients at any age and is most commonly diagnosed among adults in their 30s and 40s.1
All individuals in the posted videos have been compensated by Vera Therapeutics for their time.
Watch Dr Cheung discuss the impact of IgAN on kidney function1 in the above video and see more expert-led videos here.
Most patients are diagnosed at stage 3 chronic kidney disease (CKD) and often progress to ESKD in as few as 10 years from diagnosis1,4
AT DIAGNOSIS,
~75%
of patients had stage 3 CKD or higher4*
WITHIN 20 YEARS,
~75%
of patients develop kidney failure1
WITHIN A LIFETIME,
~100%
of patients remain at risk of kidney failure1,5
*In a retrospective cohort study of US patients with IgAN.4
At diagnosis, patients have already lost more than 50% of their nephrons5
Most patients have already experienced significant nephron loss by the time of diagnosis. Preserving all remaining nephrons is critical to avoid kidney failure during a patient’s lifetime.5
IgAN is associated with 4 disease markers: eGFR (kidney function),
Gd-IgA1, hematuria, and proteinuria3
These 4 disease markers are associated with poor clinical outcomes and increased risk of ESKD.3
Unless eGFR can be minimized to <1 mL/min/year, the majority of patients are at risk of ESKD in their lifetime. Proteinuria and eGFR correlate with disease severity in patients with IgAN, and minimizing eGFR decline can decrease the lifetime risk of dialysis, transplant, or death1,6
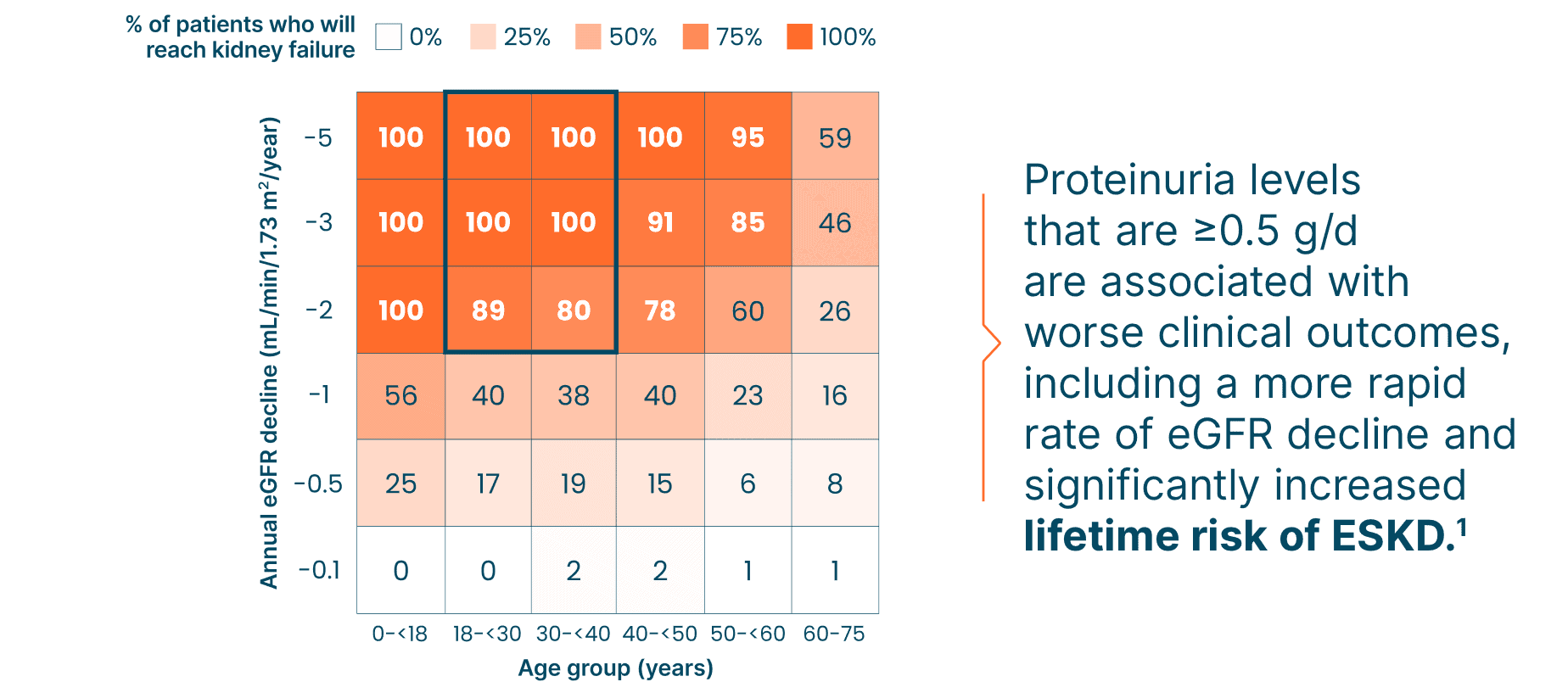
Black box highlights the most common age and annual eGFR decline at diagnosis for patients with IgAN.
Adapted from Pitcher D et al. Clin J Am Soc Nephrol. 2023;18(6):727-738. doi:10.2215/CJN.0000000000000135
In IgAN, BAFF and APRIL fuel B cells to produce a form of IgA called Gd-IgA1 that acts as an autoantigen that triggers the production of autoantibodies and formation of immune complexes, which lead to irreversible progressive kidney damage.3
Reduction or prevention of IgA immune complex formation should optimally incorporate treatments that have been proven to reduce Gd-IgA1.6,7
Gd-IgA1 and related immune complex levels correlate with proteinuria levels in patients with IgAN3,8
Adapted from Suzuki Y et al. Clin Exp Nephrol. 2014;18(5):770-777. doi:10.1007/s10157-013-0921-6
Evidence from recent studies shows that glomerular inflammation in IgAN can lead to hematuria, which is linked to a higher risk of kidney disease progression and ESKD.9-11
Microscopic hematuria is the most common clinical feature of IgAN.9,10
Proteinuria levels that are ≥0.5 g/d are associated with worse clinical outcomes, including a more rapid rate of eGFR decline and significantly increased lifetime risk of ESKD.1
Evidence supports seeking maintenance of proteinuria levels <0.5 g/d (preferably <0.3 g/d).6,7
Even “low” levels of proteinuria are associated with high risk of kidney failure or death1,5
These “low” levels could leave patients at risk of kidney failure in as few as 10 years after diagnosis1,5
To mitigate the risk of ESKD, it may be time to move beyond supportive care alone and concomitantly target IgAN at the source with disease-modifying treatment.6
BAFF and APRIL: 2 drivers of IgAN disease progression3
Take a look at clinical and preclinical evidence for the dual role of BAFF and APRIL in IgAN pathophysiology, and see how their upregulation initiates the multi-hit model that leads to negative outcomes.
Discover the latest peer-reviewed research and expert insights
Find publications and expert-led videos about IgAN, including the role of BAFF and APRIL and other key topics.
Keep up with the latest advancements in IgAN
Sign up to receive emails on all the latest IgAN insights, news, and research from Vera Therapeutics. These updates are intended for US healthcare professionals only.
References: 1. Pitcher D, Braddon F, Hendry B, et al. Long-term outcomes in IgA nephropathy. Clin J Am Soc Nephrol. 2023;18(6):727-738. doi:10.2215/CJN.0000000000000135 2. Kwon CS, Daniele P, Forsythe A, Ngai C. A systematic literature review of the epidemiology, health-related quality of life impact, and economic burden of immunoglobulin A nephropathy. J Health Econ Outcomes Res. 2021;8(2):36-45. doi:10.36469/001c.26129 3. Cheung CK, Barratt J, Liew A, Zhang H, Tesar V, Lafayette R. The role of BAFF and APRIL in IgA nephropathy: pathogenic mechanisms and targeted therapies. Front Nephrol. 2024;3:1346769. doi:10.3389/fneph.2023.1346769 4. Caster DJ, Abner CW, Walker PD, et al. Clinicopathological characteristics of adult IgA nephropathy in the United States. Kidney Int Rep. 2023;8(9):1792-1800. doi:10.1016/j.ekir.2023.06.016. Erratum in: Kidney Int Rep. 2023;8(12):2842. doi:10.1016/j.ekir.2023.10.002 5. Barratt J, Lafayette RA, Floege J. Therapy of IgA nephropathy: time for a paradigm change. Front Med (Lausanne). 2024;11:1461879. doi:10.3389/fmed.2024.1461879 6. Floege J, Bernier-Jean A, Barratt J, Rovin B. Treatment of patients with IgA nephropathy: a call for a new paradigm. Kidney Int. 2025;107(4):640-651. doi:10.1016/j.kint.2025.01.014 7. Glassock RJ. An expert opinion on current and future treatment approaches in IgA nephropathy. Adv Ther. 2025;42(6):2545-2558. doi:10.1007/s12325-025-03187-7 8. Suzuki Y, Matsuzaki K, Suzuki H, et al. Serum levels of galactose-deficient immunoglobulin (Ig) A1 and related immune complex are associated with disease activity of IgA nephropathy. Clin Exp Nephrol. 2014;18(5):770-777. doi:10.1007/s10157-013-0921-6 9. Yu GZ, Guo L, Dong JF, et al. Persistent hematuria and kidney disease progression in IgA nephropathy: a cohort study. Am J Kidney Dis. 2020;76(1):90-99. doi:10.1053/j.ajkd.2019.11.008 10. Sevillano AM, Gutiérrez E, Yuste C, et al. Remission of hematuria improves renal survival in IgA nephropathy. J Am Soc Nephrol. 2017;28(10):3089-3099. doi:10.1681/ASN.2017010108 11. Zand L, Fervenza FC, Coppo R. Microscopic hematuria as a risk factor for IgAN progression: considering this biomarker in selecting and monitoring patients. Clin Kidney J. 2023;16(Suppl 2):ii19-ii27. doi:10.1093/ckj/sfad232